 |
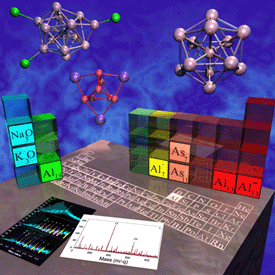 Superatoms Superatoms
Atomic Clusters,
agglomerations of a few to a few hundred atoms which are bound by
delocalized bonding, exhibit size dependent properties which vary
markedly depending on the size, shape, or composition. The stability of
clusters may be understood within a variety of models, including the Jellium Model, Aromaticity, and Wade-Mingos Rules,
depending on the geometry and metallicity of the the cluster. Because
certain cluster sizes and electron counts are quite stable relative to
others of similar size, these clusters' physical and chemical
properties are dominated by their desire to reach a certain valence
state. These clusters who have a strong propensity towards a well
defined valence are called superatoms
because they exhibit tendencies reminscent of an atom or group of the
periodic table.
Al13
is one example of a superatom cluster. Al13 has
39 valence electrons, and within the jellium
model, clusters with 40 electrons have large HOMO-LUMO Gaps and
enhanced stability. Because of this, the cluster has a very high
electron affinity of 3.4 eV, and the anion has a large HOMO-LUMO Gap of
1.87 eV. Because the chemical and electrostatic properties are
dominated by the enhanced stability it recieves by moving to a valence
of -1, 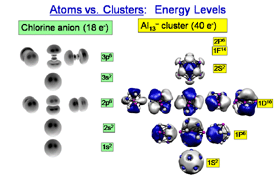 the Al13 cluster is
considered a Superhalogen Superatom.
While the superatom behavior is determined by the electronic levels
near the Fermi energy, in simple metal clusters the entire valence
electronic structure is made of electronic shells, which are multiple
highly degenerate states, much like atoms. For such clusters, the jellium model
offers a simplified framework for explaining the observed behavior.
Employing an extension of the independent nuclear shell model to a
simple electron gas concept developed by Cohen and Chou, Knight and
co-workers accounted for the observations. In this model, one imagines
that the positive charge of all of the ions in the cluster is
distributed uniformly over a sphere of the size of the cluster. The
energy levels of electrons for such a charge distribution correspond to
1s2 1p6 1d10
2s2 1f14 2p6,
The magic sizes thus correspond to filled
electronic shells, suggesting the role of these on the stability. the Al13 cluster is
considered a Superhalogen Superatom.
While the superatom behavior is determined by the electronic levels
near the Fermi energy, in simple metal clusters the entire valence
electronic structure is made of electronic shells, which are multiple
highly degenerate states, much like atoms. For such clusters, the jellium model
offers a simplified framework for explaining the observed behavior.
Employing an extension of the independent nuclear shell model to a
simple electron gas concept developed by Cohen and Chou, Knight and
co-workers accounted for the observations. In this model, one imagines
that the positive charge of all of the ions in the cluster is
distributed uniformly over a sphere of the size of the cluster. The
energy levels of electrons for such a charge distribution correspond to
1s2 1p6 1d10
2s2 1f14 2p6,
The magic sizes thus correspond to filled
electronic shells, suggesting the role of these on the stability.
The
next chapter in superatoms arrived with the identification of
Aluminum-Halide superatoms in the gas phase. It was found that while
other cluster species  readily reacted away, Al13I-
and Al13I2-,
which bear direct resemblance to well-known di- and tri-halide ions,
were quite resistant to etching. Theoretical studies on Al13I-
showed that the clusters stability resides in the ability of the Al13
moiety to retain its anionic characteristics, in terms of both geometry
and charge state, even in the presence of an iodine atom. The aluminum
cluster actually pulls charge away from the iodine atom. The discovery
and characterization of Al13I-
and Al13I2-
showed that Al13 can truly be described as a
superhalogen (recall that stable X2-
and X3- complexes, where
X denotes a halogen, are well-characterized). Additional work on the Al14Ix-
clusters revealed that the Al14 also seeks a
valence state of +2, which also corresponds to the 40 e-
shell closing. Al7- was
found to have Multiple Valence in
which it may tend towards both +2 and +4 states depending on the
reactant. Superatom have been identified experimentally in the gas
phase using Oxygen Etching, as
cluster with closed electronic shells are inert when
exposed to Oxygen
at
room temperature, while open shell clusters react readily. readily reacted away, Al13I-
and Al13I2-,
which bear direct resemblance to well-known di- and tri-halide ions,
were quite resistant to etching. Theoretical studies on Al13I-
showed that the clusters stability resides in the ability of the Al13
moiety to retain its anionic characteristics, in terms of both geometry
and charge state, even in the presence of an iodine atom. The aluminum
cluster actually pulls charge away from the iodine atom. The discovery
and characterization of Al13I-
and Al13I2-
showed that Al13 can truly be described as a
superhalogen (recall that stable X2-
and X3- complexes, where
X denotes a halogen, are well-characterized). Additional work on the Al14Ix-
clusters revealed that the Al14 also seeks a
valence state of +2, which also corresponds to the 40 e-
shell closing. Al7- was
found to have Multiple Valence in
which it may tend towards both +2 and +4 states depending on the
reactant. Superatom have been identified experimentally in the gas
phase using Oxygen Etching, as
cluster with closed electronic shells are inert when
exposed to Oxygen
at
room temperature, while open shell clusters react readily.
Work
in other groups, including Prof. Whetten, Prof. Hakkinen, Prof. Murray, and
Prof. Kornberg among
others have studied a variety of gold based cluster assembled materials
which are stabilized by thiol based ligands. The stability of the
clusters which are cystallized are derived from the jellium model, in
which the electrons from gold, and the electrons withdrawn by the
electronegative linkers are considered.
-
1) S.N. Khanna, P.Jena. "Atomic Clusters:
Buidling Blocks for a New Class of Solids" Phys. Rev. B 51 13705 (1995).
2) D.E. Bergeron, A.W. Castleman Jr., T.
Morisato, and S.N. Khanna. "Formation of Al13I-:
Evidence for the superhalogen character of Al13-."
Science 304 84 (2004).
3) D.E. Bergeron, P.J. Roach, A.W.
Castleman Jr., N. Jones, and S.N. Khanna. "Al Cluster Superatoms as
Halogens in Polyhalides and as Alkaline Earths in Iodide Salts "
Science 307 231 (2005).
4) J.U. Reveles, S.N. Khanna, P.J. Roach,
and A.W. Castleman Jr. "Multiple valence superatoms " Proc. Nat. Acad.
Sci. 103 18405 (2006).
5) A.C. Reber, S.N. Khanna, and A.W.
Castleman Jr. "Superatom Compounds, Clusters, and Assemblies: Ultra
Alkali Motifs and Architectures. " J. Amer. Chem Soc. 129 10189 (2007).
6) A.C. Reber, S.N. Khanna, P.J. Roach,
W.H. Woodward, and A.W. Castleman Jr. "Spin Accomodation and Reactivity
of Aluminum Based Clusters with O2" J. Amer.
Chem Soc. 129 16098 (2007).
7) P.D. Jadzinsky, G. Calero, C.J.
Ackerson, D.A. Bushnell and R.D. Kornberg. (2007)." Structure of a
thiol monolayer-protected gold nanoparticle at 1.1Å resolution."
Science 318 5149 (2007)
8) M. Walter, "A unified view of
ligand-protected gold clusters as superatom complexes" Proc. Nat. Acad.
U.S.A. 105 9157 (2008).
9) A.W. Castleman Jr., and S.N. Khanna.
"Clusters, Superatoms, and Building Blocks of New Materials." J. Phys.
Chem. C 113 2664.
|
|

